Overview
In cooking, brining is a process similar to marination in which meat is soaked in a salt solution (the brine) before cooking. Brining makes cooked meat moister by hydrating the cells of its muscle tissue before cooking and by allowing the cells to hold on to the water while they are cooked,
The brine surrounding the muscle fiber cell has a higher concentration of salt than the fluid within the cells. This leads salt ions to enter the cell via diffusion. The high salt concentration immediately begins to do its work on the protein complexes within the muscle fiber (see below for detailed explanation). The end result is the muscle fibers draws in and retain a substantial amount of water by both osmosis and capillary action. The meat’s weight can increases by 10% or more, allowing for greater moisture in the food after cooking. In addition, the dissolved protein does not coagulate into the normally dense aggregates, so the cooked meat seems more tender.
Changes in Muscle Fiber as a Result of Brining
The main structural component of meat is the myofibril which occupies about 70% percent of the volume of lean meat. Mybofibrils contain about 20% of protein, the rest being water. Thus the majority of water in meat lies in the myofibrils in the spaces between the thick and thin filaments. Myofibrils can swell to more than twice their original volume when immersed in salt solutions (Offer and Trinick, 1983) .
In porcine meat three distinct water populations were identified using proton LF NMR: one tightly bound to the the muscle proteins; a second trapped within the myofibril structure; and the third corresponding to water outside the myofibrillar lattice or even outside the muscle cells (Bertram et. al. 2001).
In meat water seems to be held by capillary action. The majority in the interfilament spaces with the myofribrils but a substantial part in extracellular space and the spaces between myofibrils. From Offer: the major effect on the increase in water mobility was probably due to the increased protein electrostatic repulsion leading to increase myofilament spacing. at a concentration of 4.6 - 5.8 %
| Changes in myofibrils in high salt solutions have been verified using phase contrast microscopy (Offer and Trinick, 1983; and scanning electron microscopy Graiver et. Al. 2006). At low NaCl concentrations, swelling of the fibers, and high values of water holding capacity were observed by Offer and Trinick (1983); Belitz and Grosch (1997) and Gravier et.al (2005). The increase in water holding capacity is likely attributed to the lateral expansion of myofibrils, which is coupled to protein solubilization (see below). |
While low concentrations of NaCl produced swelling of the fibers, and high values of the water holding capacity at higher NaCl concentrations the phenomenon was reversed, fiber volume decreased, the tissue lost its own water and proteins precipitated causing disruption in the matrix Gravier et.al (2005).
To understand the mechanism further it is important to first take a look at muscle fiber anatomy.
The basic unit of the muscle fiber is the sarcomere.
A sarcomere is the basic unit of a muscle's cross-striated myofibril. Sarcomeres are multi-protein complexes composed of three different filament systems. Sarcomeres are multi-protein complexes composed of three different filament systems.
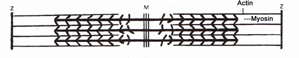
MUSCLE FIBER AT BIOLOGICAL SALT CONCENTRATIONS |
The thick filament system is composed ofmyosin protein which is connected from the M-line to the Z-disc by Titin It also contains myosin-binding protein C which binds at one end to the thick filament and the other to Actin. The thin filaments are assembled by actinmonomers bound to Nebulin. Which also involves tropomyosin; a dimer which coils itself around the F-actin core of the thin filament. In a sarcomere under physiological salt conditions, the thick and thin filaments are tightly held in place. |
Mechanism of Brining at muscle fiber level -- thick and thin fibers repel
McGee in his text "On Food and Cooking', pg 155-156 states that: “Brining has two initial effects. First, salt disrupts the structure of the muscle filaments. A 3 percent salt solution (2 tablespoons per quart/30 gm. per liter) dissolves parts of the protein structure that supports the contracting filaments themselves. "Second, the interactions of salt and proteins result in a greater water-holding capacity in the muscle cells, which then absorb water from the brine…The meat’s weight increases by 10% or more…In addition, the dissolved protein filaments can’t coagulate into the normally dense aggregates, so the cooked meat seems more tender.” (pages 155-156) .
Olfer and Trinick (1983) explain that it is primarily the chloride ions in salt (NaCl) binding to the filaments that allows for the filament lattice expansion to occur (see below).
Myofibrils have been observed by phase contrast microscopy, and are seen to swell quickly to about twice their original volume in salt solutions resembling those used in meat processing. Such swelling is highly cooperative. Pyrophosphate reduces very substantially the sodium chloride concentration required for maximum swelling. In the absence of pyrophosphate, swelling is accompanied by extraction of the middle of the A-band; in its presence the ,4-band is completely extracted, beginning from its ends. Offer and Trinick suggest that that CI- ions bind to the filaments and increase the electrostatic repulsive force between them. A crucial factor in swelling is likely to be the removal at a critical salt concentration of one or more transverse structural constraints in the myofibril (probably crossbridges, the M-line or the Z-line) allowing the filament lattice to expand.
As long as the cross-bridges remain attached the lattice cannot swell much: conversely, if the lattice does swell appreciably, the cross-bridges cannot remain attached. Under such circumstances one can see why the swelling should be a highly cooperative phenomenon: when the cross-bridges dissociate they must all do so at the same time to allow swelling. When this occurs the thick filament backbone will no longer be stabilized and depolymerisationoccurs from the ends as with separated thick filaments.
A final concentration of 0-8 to 1M (4-6-5.8%) sodium chloride gives maximum water uptake Xiong et.al (2000). Salting out occurs at about 90-100 g/l At higher salt concentrations a reverse effect was observed. Fiber volume decreased, the tissue lost its own water and proteins precipitated causing disruption in the matrix.
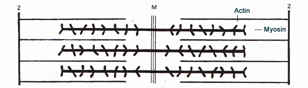
MUSCLE FIBER IN PRESENCE OF HIGH SALT CONCENTRATION -- NOTE LATTICE EXPANSION |
Chloride ions bind to the filaments and increase electrostatic repulsive forces between them. A crucial factor in swelling is likely to be removal at a critical salt concentration of one or more transverse structural constraints in the myofybril. allowing the filament lattice to expand. The attached cross-bridges together with the ZM-Z- lines resist swelling Reference: Offer G and Trinick J, (1983) On the Mechanism of Water Holding in Meat, Meat Science 8: 245-281. |
The sort of salt concentration needed to see the dissociation effect biochemically is typically around 500mM salt concentration (150mM being roughly physiological). 150g salt in 1 gallon (3.78 L) is 680 mM. This converts to 5.3 oz salt /gallon of water.
It seems reasonable to suppose that water is held in meat by capillarity, the majority in the interfilament spaces within the myofibrils, but a substantial part in the extracellular space and the spaces between myofibrils. Offer G and Trinick J, (1983)
STANDARD BRINING TABLE:
| TYPE OF BRINE | COLD WATER | SALT | SUGAR | AMOUNT OF BRINE | TIME |
| BASIC | 1 Quart | 1/2 cup Diamond Crystal Kosher 1/4 cup + 2 Tablsp Morton Kosher 1/4 cup table salt Any by Weight = 5 oz. of salt |
1/2 cup | 1 quart per pound of food not to exceed 2 gallons of brine | 1 hr per pound but not less than 3 hrs. or more than 8 hours |
| HIGH HEAT ROASTING OR GRILLING | 1 Quart | 1/4 cup Diamond Kosher 3 tablespoons Morton Kosher 2 Tablespoons table |
2 tablsp. | 1 quart per poound of food, not to exceed 2 gallons of brine | 1 hour per pound but not less than 30 min or more than 8 hours. |
| Reference: Cooks Illustrated -- Basics of Brining -- See below for other Brining Solutions | |||||
HEALTH NOTE: Make sure the brining solution has been sufficiently chilled before adding your meat. Always leave the brine in the refrigerator.
Comparing Brining Solutions
ARE YOU USING TOO MUCH SALT IN YOUR BRINE?
When it comes to brining food it is best to start with lower concentrations and/or brining times.
To get a better perspective of salt brines keep in mind that the salinity of fish and meat is around 9g/L which converts to a 0.9% salt solution.
On average, seawater in the world's oceans has a salinity of ~3.5%, or 35 parts per thousand. This means that every 1 kg of seawater has approximately 35 grams of dissolved salts (mostly, but not entirely, the ions of sodium chloride: Na+, Cl-). This approximates at 35 gm of salt/ 1 Liter of water.
The above brine table recommended by Cooks Illustrated contains 5 oz. of salt / quart of water:
5 oz. = 142 gms. / 0.909 L = 156.2 gm/L or a 15% brine solution.
Dr. Estes Reynolds, a brining expert at the University of Georgia. (see article by Shirley Corriher in References) suggests using around half the amount of salt as the Cooks Illustrated recipe or 9.6 ounces of salt (272 grams) for every gallon of water.1 US gallon = 3.78541178 liters
272 GMs/ 3.785 l = 72g/l >>> twice that of sea water.... or 7.2 % brine
Keep in mind that 4-6-5.8% sodium chloride gives maximum water uptake in muscle fiber (Xiong et.al 2000). In addition many protein changes are also seen at these salt concentrations. As pointed about by McGee (On Food and Cooking) a 3 percent salt solution (2 tablespoons per quart/30 gm. per liter) dissolves parts of the protein structure that supports the contracting filaments themselves.
Loss of water during cooking
Even brining will not prevent water loss due to shrinkage during cooking, although since we are starting with more water --and the change in protein structure may make water expulsion more difficult -- loss of water is inevitable when cooking occurs at high temperatures.
Bendall and Restall (1983) observed that the shrinkage rate was maximal at 60 °C and that a slow but significant shrinkage of the fibres occurred at 40°C. The melting temperature of fibrous collagen is substantially higher (about 60-65 °C)
When single myofibres are heated in an aqueous medium up to temperatures of 90°C at pH 5·5, they do not shorten but instead decrease in diameter. This decrease begins slowly at 40° and reaches a maximal rate and extent at 60°, when the myofibre volume has decreased to 50% of the initial volume and about 60% of the cell water has been expelled. Expulsion of water from the myofibre is slow and incomplete from 40 to 52.5 °, but accelerates markedly to maximal rate between 57.5 and 60 °. The only muscle protein component so far identified which would on denaturation and shrinkage demonstrate this is the Type IV/V collagen.
It is the high tension which this collagen develops during heat shrinkage which is the main cause of extrusion of fluid from the meat.
SEE ALSO: IMPORTANT TEMPERATURES IN COOKING AND SOUS VIDE COOKING
Vacuum Brining
If you want to speed up the brining process (or use less salt), some reports suggest use a Foodsaver (those vacuum machines with which you can make vacuum sealed bags or remove the air from containers.) Vacuum brining (PVB) of meat products in salt-concentrated solutions gives rise to hydrodynamic mechanisms that facilitate solution infiltration into the meat structure. See Abstract
References:
Bertram et al. (2001) H.C. Bertram, A.H. Karlsson, M. Rasmussen, O.D. Pedersen, S. Døntrup and H.J. Andersen, Origin of multiexponential T2 relaxation in muscle myowater, Journal of Agricultural and Food Chemistry 49 (2001), pp. 3092–3100.
Belitz and Grosch, 1997 H.D. Belitz and W. Grosch, Química de los alimentos (second ed.), Ed. Acribia Zaragoza, España (1997).
Bendall, J. R. (1954). J. Sci. Fd. Agric., 5, 468.
Bendall J and R,Restal D (1983) The cooking of single myofibres, small myofibre bundles and muscle strips from beef M. psoas and M. sternomandibularis muscles at varying heating rates and temperatures. J.Meat Sci, 93-117.
Graiver, N, A. Pinotti, A. Califano and N. Zaritzky. Diffusion of sodium chloride in pork tissue, Journal of Food Engineering, 77 (2006) pp. 910-918.
Hamm 1960)-- Advances in Food Research 10:355.
R. Lakshmanana, 1, John A. Parkinsonb and John R. Piggott High-pressure processing and water-holding capacity of fresh and cold-smoked salmon (Salmo salar) Food Science and Technology. 40, 3, April (2007), Pages 544-551
Morrisey et al., 1987 P. Morrisey, D. Mulvihill and E. O’Neill, Functional properties of muscle proteins. In: B.J.F. Hudson, Editor, Developments in food proteins-5, Elsevier, London and New York (1987), pp. 195–256.
G. Offer and J. Trinick, On the mechanism of water holding in meat: the swelling and shrinking of myofibrils, Meat Science 8 (1983), pp. 245–281
Y. Xiong, X. Lou, R. Harmon, C. Wang and W. Moody, Salt-and pyrophosphate-induced structural changes in myofibrils from chicken red and white muscle, Journal of Science and Food Agriculture 80 (2000), pp. 1176–1182.
Selected Reading:
Basics of Brining by Cooks Illustrated
McGee, Harold, On Food and Cooking
Wolke, Robert, What Einstein Told his Cook
Preparation of Salt Brines for the Fishing Industry
To Brine or not to Brine Part I --Craig Shelton
To Brine or not to Brine Part II --Craig Shelton
Why Brining Keeps Meat So Moist --Shirley Corriher
Here is the recipe for Alice Waters' turkey brine: Note this brine recipe uses half the amount of salt than that called for in most standard brines, but is in line with that of Reynolds (see above comments)
3/4 cup plus 2 tbsp kosher salt
3/4 cup sugar
1 carrot peeled and diced
1 large onion peeled and diced
1 leek, cleaned and sliced
2 bay leaves
1 tbsp black peppercorns
1 tbsp coriander seeds
1/4 tsp red pepper flakes
1/4 tsp fennel seeds
2 star anise
2-3 sprigs fresh thyme
In a large stockpot (16 quart or more) bring 2 gallons of water to a boil.
Add salt and sugar and stir until dissolved.Turn off heat and add veggies, then herbs and spices. Refrigerate till cold. Remove giblets from turkey. Add turkey to stock pot. Weigh down with a plate if neccessary to keep turkey below the brine's surface. Refrigerate 72 hours, then remove from brine and allow turkey to come to foom temperature. The recipe calls for 12-14 lb turkey. Ref... posted by emilief Nov 23, 2007 on chowhoud.com
Glossary of Terms:
Osmosis --Osmosis is the diffusion of water through a cell wall or membrane or any partially-permeable barrier from a solution of low solute concentration to a solution with high solute concentration, up a solute concentration gradient. It is a physical process in which a solvent moves, without input of energy, across a semi-permeable membrane (permeable to the solvent, but not the solute) separating two solutions of different concentrations.[1] Osmosis releases energy, and can be made to do work, as when a growing tree-root splits a stone. Shot of a computer simulation of the process of osmosisNet movement of solvent is from the less-concentrated (hypotonic) to the more-concentrated (hypertonic) solution, which tends to reduce the difference in concentrations.
Diffusion -- Diffusion is the movement of particles from an area of high concentration to an area of low concentration in a given volume of fluid (either liquid or gas) down the concentration gradient. For example, diffusing molecules will move randomly between areas of high and low concentration but because there are more molecules in the high concentration region, more molecules will leave the high concentration region than the low concentration one.
Capillary Action Capillary action, capillarity, capillary motion, or wicking is the ability of a substance to draw another substance into it. The standard reference is to a tube in plants but can be seen readily with porous paper. It occurs when the adhesive intermolecular forces between the liquid and a substance are stronger than the cohesive intermolecular forces inside the liquid.
Denaturation -Denaturation is a major change in protein or nucleic acid structure by application of some external stress or compound for example, treatment of proteins with strong acids or bases, high concentrations of inorganic salts, organic solvents (e.g., alcohol or chloroform), or heat.
Depolymerization --to decompose (macromolecules) into simpler compounds (as monomers)