Myristic Acid
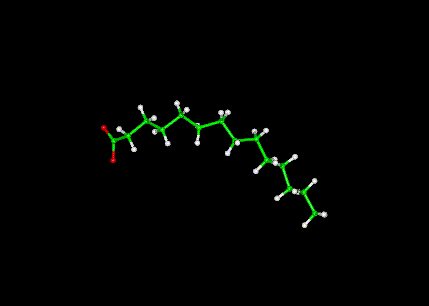
The lipid shown here is Myristic Acid in a ball-and-stick configuration. The coordinates from from the Protein Data Base--ID- 1ICM (See Myristate ligand bound to protein here.)
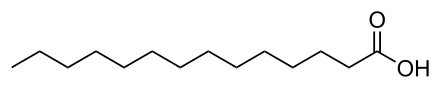
Myristic Acid ( tetradecanoic acid): is a saturated fatty acid.
Chemical Structure - CH3(CH2)12COOH - (C:D = 14:0)
The structure of saturated and unsaturated chemical bonds looks like the diagram below.
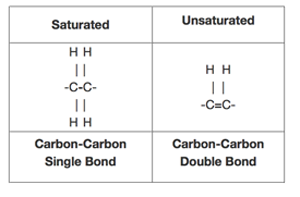
Saturated fatty acids are of the form R-COOH
where R = CH 3-(CH2)n - n being any integer greater than or equal to 1In chemistry, especially biochemistry, a fatty acid is a carboxylic acid (or organic acid), often with a long aliphatic tail (long chains), either saturated or unsaturated. Most of the natural fatty acids have an even number of carbon atoms, because they are made up of acetate which has two carbon atoms.
Industrially, fatty acids are produced by the hydrolysis of the ester linkages in a fat or biological oil (both of which are triglycerides), with the removal of glycerol.
Many Americans consume little myristic acid because this fatty acid is found by coconut oil and dairy fats that many American don't eat, Myristic acid is an important fatty acid, which the body uses to stabilize different proteins, including proteins used in the immune system.
What foods contain Myristic Acid
Nutmeg butter has 75% trimyristin, the triglyceride of myristic acid. Besides nutmeg, myristic acid is 16% of coconut oil and palm kernel oil, 7-12% of butter fat, 2-4% of beef and lamb fat, 3% of salmon fat, 2% of lard, and less than 1% of chicken.
Myristic Acid and Cholesterol Levels
Myristic acid (C14:0), apparently raises LDL cholesterol concentrations somewhat more than does palmitic acid, which is the predominant saturated fatty acid in most diets (See: CHOLESTEROL | Factors Determining Blood Cholesterol Levels)
Fats in Cooking and Health
- What are the differrent types of fatty acids?
- What are Trans Fats?
- What is the difference between LDL and HDL?
- What are the different types of omega 3 fatty acid?
- What are omega-6 fatty acids?
- The importance of the ratio of omega-6/omega-3 essential fatty acids
- What is the difference between saturated and unsaturated fats?
- What causes oils to go rancid?
- What are essential fatty acids?
- Duck Fat as an alternative to butter
- How to render duck fat
- Did butter get a bad rap?
Types of Fatty Acids
Saturated