Palmitic Acid
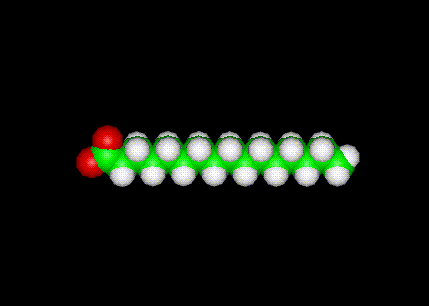
The lipid shown here is Palmitic Acid in a ball-and-stick configuration.
PalmiticAcid is a saturated fatty acid.
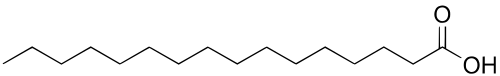
Palmitic Acid - CH3(CH2)14COOH- (C:D = 16:0)
The structure of saturated and unsaturated chemical bonds looks like the diagram below.
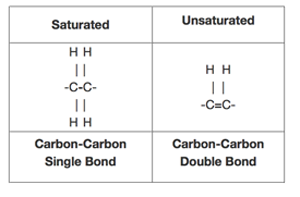
Saturated fatty acids are of the form R-COOH
where R = CH 3-(CH2)n - n being any integer greater than or equal to 1In chemistry, especially biochemistry, a fatty acid is a carboxylic acid (or organic acid), often with a long aliphatic tail (long chains), either saturated or unsaturated. Most of the natural fatty acids have an even number of carbon atoms, because they are made up of acetate which has two carbon atoms.
Palmitic acid, also called hexadecanoic acid, is one of the most common saturated fatty acids found in animals and plants. It is a white solid that melts at 63.1°C and its chemical formula is CH3(CH2)14COOH. As its name tells us, it is found in palm oil but also in butter, cheese, milk and meat.
Palmitate is a term for the salts or esters of palmitic acid. The palmitate anion is the observed form of palmitic acid at physiological pH. Palmitic acid is the first fatty acid produced during lipogenesis (fatty acid synthesis) and from which longer fatty acids can be produced. Palmitate negatively feeds back on acetyl-CoA carboxylase (ACC) which is responsible for converting acetyl-ACP to malonyl-ACP on the growing acyl chain, thus preventing further palmitate generation. Palmitate is an antioxidant and a vitamin A compound added to low fat milk to replace the vitamin content lost through the removal of milk fat. Palmitate is attached to the alcohol form of vitamin A, retinol, in order to make vitamin A stable in milk. Derivatives of palmitic acid were used in World War II to produce napalm. Reduction of palmitic acid yields cetyl alcohol.
Palmitic acid is the first fatty acid produced during lipogenesis and from which longer fatty acids can be produced.
| Palmitic acid | |
|---|---|
| Chemical name | Hexadecanoic acid |
| Other names | Palmitic acid hexadecylic acid cetylic acid |
| Chemical formula | C16H32O2 |
| Molecular mass | 256.42 g/mol |
| CAS number | [57-10-3] |
| Density | 0.853 g/cm3 at 62 °C |
| Melting point | 63-64 °C |
| Boiling point | 215 °C at 15 mmHg |
| SMILES | CCCCCCCCCCCCCCCC(=O)O |
Where is Palmitic Acid Found?
Palmitic acid is found naturally in palm oil and palm kernel oil, as well as in butter, cheese, milk and meat. Fatty cuts of red meat, as well as skin-on poultry, contain relatively high levels of saturated fat and contain large amounts of palmitic acid.
Palmitic Acid and Health
from Palmitic Acid and Health (2016)"...Interest in the dietary role and metabolic effect of saturated fatty acids has been recently renewed on the basis of epidemiologic observations and economical approach to health and well-being. Saturated fats may favorably increase blood HDL-Cholesterol levels without significant changes of the total cholesterol/HDL-Cholesterol ratio. Also, the negative effect of saturated fat on cardiovascular diseases risk has recently been challenged. Palmitic acid, among all, may have special structural and functional roles in utero and in infancy, and indeed is it is being delivered in a unique form in human milk. Future research should include objective cost-benefit analyses when disentangling the role of saturated fats in dietary recommendations..."
Fats in Cooking and Health
- What are the differrent types of fatty acids?
- What are Trans Fats?
- What is the difference between LDL and HDL?
- What are the different types of omega 3 fatty acid?
- What are omega-6 fatty acids?
- The importance of the ratio of omega-6/omega-3 essential fatty acids
- What is the difference between saturated and unsaturated fats?
- What causes oils to go rancid?
- What are essential fatty acids?
- Duck Fat as an alternative to butter
- How to render duck fat
- Did butter get a bad rap?
Types of Fatty Acids
Saturated