CHEMICAL AND PHYSICAL PROPERTIES OF SUCROSE
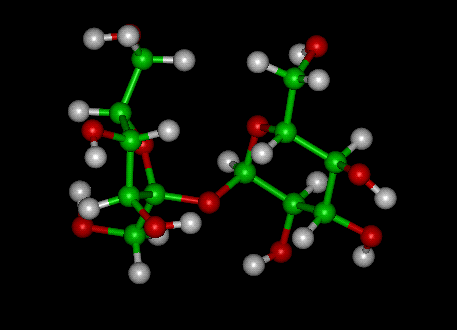 |
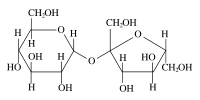
Sucrose (common name: table sugar, also called saccharose) is a disaccharide (glucose + fructose) with the molecular formula C12H22O11. Its systematic name is α-D-glucopyranosyl-(1→2)-β-D-fructofuranose. It is best known for its role in human nutrition and is formed by plants but not by higher animals.
Physical and chemical properties
Pure sucrose is most often prepared as a fine, colorless, odorless crystalline powder with a pleasing, sweet taste. Large crystals are sometimes precipitated from water solutions of sucrose onto a string (or other nucleation surface) to form rock candy, a confection.
Like other carbohydrates, sucrose has a hydrogen to oxygen ratio of 2:1. It consists of two monosaccharides, α-glucose and fructose, joined by a glycosidic bond between carbon atom 1 of the glucose unit and carbon atom 2 of the fructose unit. What is notable about sucrose is that unlike most polysaccharides, the glycosidic bond is formed between the reducing ends of both glucose and fructose, and not between the reducing end of one and the nonreducing end of the other. The effect of this inhibits further bonding to other saccharide units. Since it contains no free anomeric carbon atom, it is classified as a nonreducing sugar.
Sucrose melts and decomposes at 186 °C to form caramel, and when combusted produces carbon, carbon dioxide, and water. Water breaks down sucrose by hydrolysis, however the process is so gradual that it could sit in solution for years with negligible change. If the enzyme sucrase is added however, the reaction will proceed rapidly.
Reacting sucrose with sulfuric acid dehydrates the sucrose and forms elemental carbon, as demonstrated in the following equation:
- C12H22O11 + H2SO4 catalyst --->12 C + 11 H2O
Commercial production and use
Sucrose is the most common food sweetener in the industrialized world, although it has been replaced in industrial food production by other sweeteners such as fructose syrups or combinations of functional ingredients and high intensity sweeteners.
Sucrose is the most important sugar in plants, and can be found in the phloem sap. It is generally extracted from sugar cane or sugar beet and then purified and crystallized. Other (minor) commercial sources are sweet sorghum and sugar maples.
Sucrose is ubiquitous in food preparations due to both its sweetness and its functional properties; it is important to the structure of many foods including biscuits and cookies, ice cream and sorbets, and also assists in the preservation of foods. As such it is common in many processed and so-called junk foods.
Sugar as a macronutrient
Human beings, and in fact most other mammals except felines, which lack the ability to taste sweetness will typically accept food sweetened with sucrose even if they are not hungry. In mammals, sucrose is very readily digested in the stomach into its component sugars, by acidic hydrolysis. This step is performed by a glycoside hydrolase, which catalyzes the hydrolysis of sucrose to the monosaccharides glucose and fructose. Glucose and fructose are rapidly absorbed into the bloodstream in the small intestine. Undigested sucrose passing into the intestine is also broken down by sucrase or isomaltase glycoside hydrolases, which are located in the membrane of the microvilli lining the duodenum. These products are also transferred rapidly into the bloodstream. Sucrose is digested by the enzyme invertase in bacteria and some animals.Acidic hydrolysis can be used in laboratories to achieve the hydrolysis of sucrose into glucose and fructose.
In human nutrition
Sucrose is an easily assimilated macronutrient that provides a quick source of energy to the body, provoking a rapid rise in blood glucose upon ingestion. However, pure sucrose is not normally part of a human diet balanced for good nutrition, although it may be included sparingly to make certain foods more palatable. Overconsumption of sucrose has been linked with some adverse health effects. The most common is dental caries or tooth decay, in which oral bacteria convert sugars (including sucrose) from food into acids that attack tooth enamel. Sucrose, as a pure carbohydrate, has a high food energy content (4 kilocalories per gram or 17 kilojoules per gram), and thus can make a diet hypercaloric even in small amounts, contributing to obesity.
The rapidity with which sucrose raises blood glucose can cause problems for people suffering from defects in glucose metabolism, such as persons with hypoglycemia or diabetes mellitus. Sucrose can contribute to development of the metabolic syndrome. An experiment with rats that were fed a diet one-third of which was sucrose may serve as a model for the development of the metabolic syndrome. The sucrose first elevated blood levels of triglycerides, which induced visceral fat and ultimately resulted in insulin resistance.
References
Satoshi Fukuchi (2004). "Role of Fatty Acid Composition in the Development of Metabolic Disorders in Sucrose-Induced Obese Rats". Experimental Biology and Medicine 229 (6): 486-493. PMID 15169967.
Yudkin, J.; Edelman, J., Hough, L. (1973). Sugar - Chemical, Biological and Nutritional Aspects of Sucrose. The Butterworth Group. ISBN 0-408-70172-2.
Tastant Molecules
Sugars
Artificial Sweeteners
Bitter Tastants
Umami
Acids
Thermal
